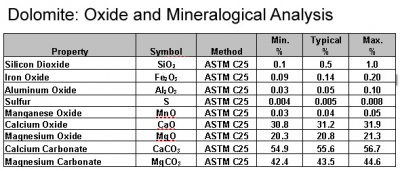
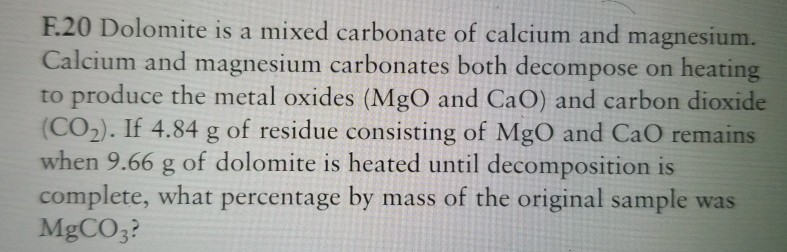![SOLVED:To find the mass percent of dolomite [CaMg(CO3)2] in a soil sample, a geochemist titrates 13.86 g of soil with 33.56 mL of 0.2516 M HCl . What is the mass percent SOLVED:To find the mass percent of dolomite [CaMg(CO3)2] in a soil sample, a geochemist titrates 13.86 g of soil with 33.56 mL of 0.2516 M HCl . What is the mass percent](https://cdn.numerade.com/previews/bf025e95-842a-4827-8af2-5174088eb353_large.jpg)
SOLVED:To find the mass percent of dolomite [CaMg(CO3)2] in a soil sample, a geochemist titrates 13.86 g of soil with 33.56 mL of 0.2516 M HCl . What is the mass percent

Best Grade Low Iron Magnesium Carbonate(dolomite) For Industrial Uses. - Buy Dolomite Agriculture,Dolomite,Magnesium Carbonate Product on Alibaba.com
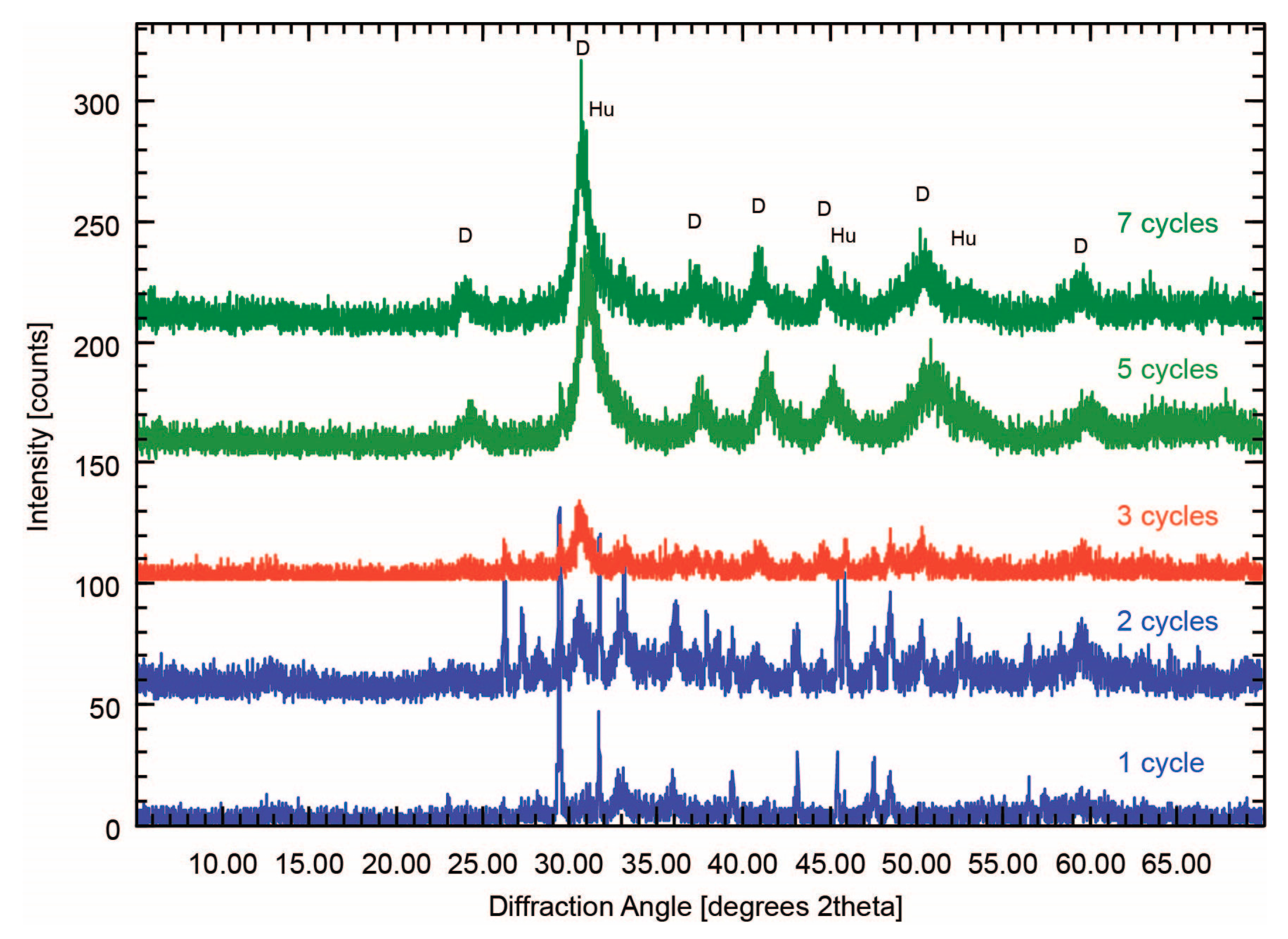
Minerals | Free Full-Text | Effect of pH Cycling and Zinc Ions on Calcium and Magnesium Carbonate Formation in Saline Fluids at Low Temperature

Classification of limestones and dolostones based on the percentage of... | Download Scientific Diagram

Dolomite Per Kg in Ethiopia (Dolostone) Calcium Magnesium Carbonate Made Pink Green White - Arad Branding

Greenhouse conditions induce mineralogical changes and dolomite accumulation in coralline algae on tropical reefs | Nature Communications

Amorphous Calcium–Magnesium Carbonate (ACMC) Accelerates Dolomitization at Room Temperature under Abiotic Conditions | Crystal Growth & Design

2.36 g of sample of dolomite containing only CaCO(3) and MgCO(3) were dissolved in 700 mL of 0.1 N HCl. The solution was dilutied to 2.05 L. 25 mL of this solution